Today NASHP’s Center for State Rx Drug Pricing celebrates five years of state action to lower drug costs. Since 2016, over a thousand new bills to constrain drug prices have been introduced across the nation, with legislative action in every state. Since 2017, forty-eight states have enacted 166 state laws to regulate pharmacy benefit managers, unlock the black box of pricing through transparency laws, enable the wholesale importation of drugs from Canada, create Prescription Drug Affordability Boards and more. Here we review a bit about NASHP’s efforts and the work supporting the state officials who have led the way.
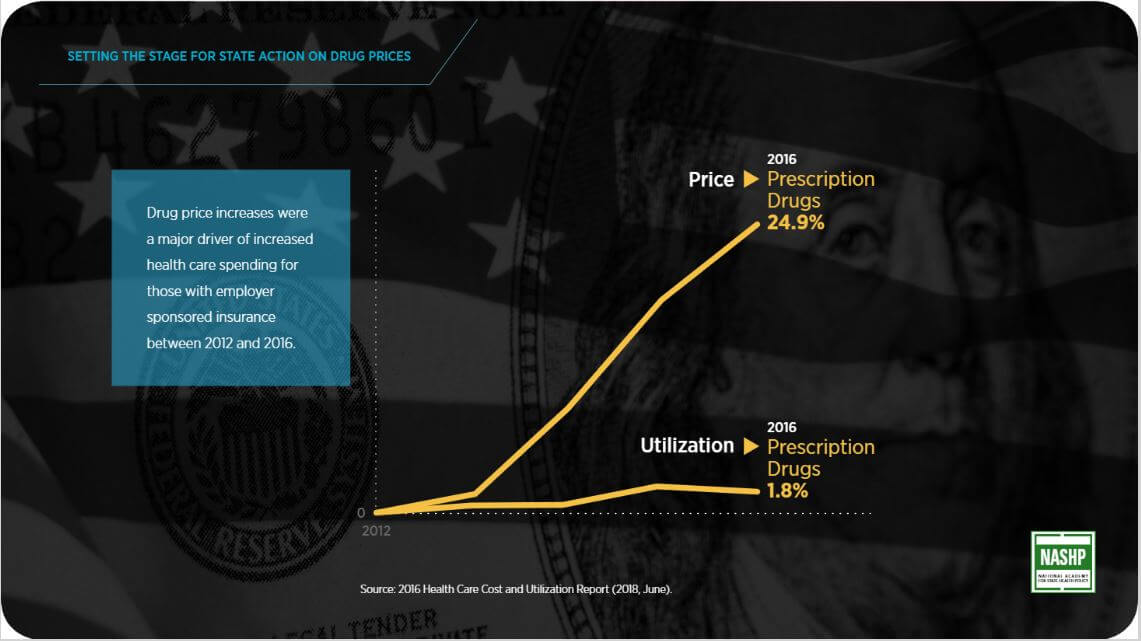
Five years ago, states felt the impact of unpredictable and persistent increases in drug prices and employers and insurance regulators were reeling as drug costs comprised about 20% of premiums. Federal action was stalled and states simply could not wait as excessive drug prices kept critically important medicines out of peoples’ reach and challenged state mandates to balance budgets.
But lowering drug costs, a priority identified by a NASHP summit of state leaders, and one often met with opposition from the powerful pharmaceutical industry, required new approaches and sustained support for states.
With a dedicated group of state officials and expert guidance from policy and legal experts, steeped in knowledge of the pharmacy supply chain, the FDA, and the legal minefields states would confront, NASHP set out to craft our first bipartisan list of actions states could take to lower prescription drug prices. To put this plan into action we launched the Center for State Rx Drug Pricing, an effort made possible thanks to the generosity of Arnold Ventures.
We established goals and strategies with policymakers to disrupt the drug supply chain and the impact of its misaligned incentives on pricing, engage states in “safe harbor” strategy discussions, and develop model legislation states could use to address drug costs. NASHP supports state bill sponsors, and – once bills are enacted – convenes state leaders to work together, with NASHP’s assistance, on effective implementation. This work has built a strong community of state leaders engaged in the common work of implementing transparency laws, drug importation and drug affordability boards.
While states are constrained by federal patent law in how far they can go, our team of lawyers helped us develop models that were within state jurisdiction and to craft approaches to anticipate potential legal challenges from the industry. We knew states would face heavy opposition and considerable odds but recognized that every legislative debate would gain considerable media, advocate, and public attention and help educate about the issues of drug pricing and the response to industry challenges. In turn, that information, and states serving as laboratories to pass new laws to constrain costs, could inform the federal debate.
State action far outstrips congressional efforts but in 2018, after 33 states had already done so, Congress enacted provisions to outlaw “gag clause” and allow pharmacists to share savings information with consumers.
The effort continues as NASHP advances new models and works with states to sustain what is now clearly an unrelenting movement among them to lower drug costs. As we celebrate five years of working with and for states to build that momentum, we anticipate more action in the years ahead. I am proud and humbled to have launched and led this effort but as I take my leave, I know that NASHP’s team – Maureen Hensley Quinn, Jennifer Reck, Johanna Butler, Sarah Lanford, Drew Gattine and Amanda Attiya – along with our incoming executive director Hemi Tewarson and our state work group, will continue to build on the considerable successes they and the states have already achieved. I look forward to reading the next five-year report!